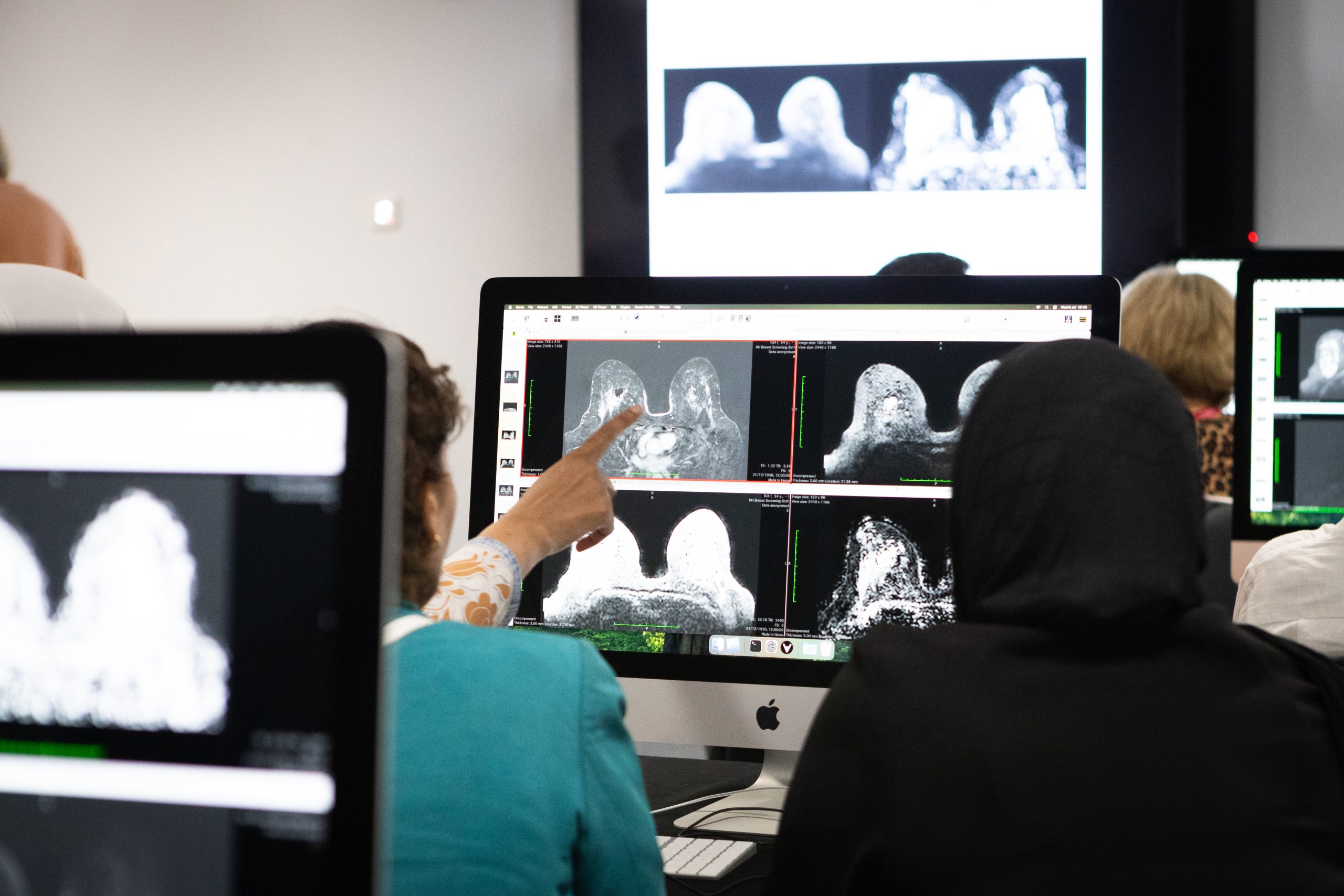
Breast imaging is rapidly evolving through AI, multimodal imaging, and workflow innovation. This article explores how clinical practice is shifting from traditional screening toward more personalised, data-driven, and integrated care models.
Breast imaging is in a period of sustained transformation. Rather than a single disruptive breakthrough, progress is being driven by the steady convergence of improved imaging technology, data integration, and artificial intelligence. The result is a field that is increasingly precise, more personalised, and more operationally complex than ever before.
At the centre of this evolution is a shift in mindset: from imaging as a largely reactive diagnostic tool to imaging as a predictive, longitudinal component of cancer risk management.
A New Baseline: Digital Maturity in Breast Imaging
Over the past decade, breast imaging has completed its transition into a fully digital ecosystem. Full-field digital mammography and digital breast tomosynthesis (DBT) are now widely established in screening and diagnostic pathways across many healthcare systems.
This digital foundation matters because it enables everything that follows. High-quality, standardised image datasets are the prerequisite for advanced analytics, AI integration, and cross-institutional learning.
However, digital maturity has also created a new challenge: volume and complexity. Radiology departments are facing rising screening demand, more incidental findings, and increasing pressure on reporting capacity. This is one of the key drivers behind current innovation.
Artificial Intelligence: From Experiment to Infrastructure
Artificial intelligence is no longer peripheral to breast imaging—it is becoming embedded within clinical workflows.
Early AI applications focused on detection support in mammography, often positioned as “second reader” systems. These tools have now evolved into broader platforms that support:
- Image triage and prioritisation
- Lesion detection and characterisation
- Breast density assessment
- Risk stratification
- Workflow optimisation in reporting environments
Importantly, AI is increasingly being evaluated not just on diagnostic accuracy, but on its impact on system performance—including reading time, recall rates, and screening efficiency.
What is emerging is a more realistic role for AI: not replacement of radiologists, but augmentation of decision-making under increasing workload pressure.
At the same time, the field is still grappling with key constraints:
- Dataset bias and representativeness
- Regulatory variability across regions
- Integration into heterogeneous clinical systems
- Clinician trust and interpretability
As a result, AI adoption is progressing unevenly—advanced in some screening programmes, still exploratory in others.
Beyond Mammography: Multimodality Is Becoming Standard
Another defining feature of current innovation is the shift toward multimodal imaging strategies.
While mammography remains the backbone of screening, there is increasing integration of:
- Breast ultrasound (including automated systems)
- Magnetic resonance imaging (MRI)
- Contrast-enhanced mammography
- Adjunct imaging in high-risk or dense breast populations
The clinical motivation is clear: no single modality is sufficient for all patients. Dense breast tissue, in particular, remains a limitation for mammography alone and has driven adoption of supplemental imaging strategies.
What is changing now is not just the availability of these modalities, but the integration of their outputs into unified diagnostic decision-making frameworks, increasingly supported by AI-driven synthesis tools.
From Population Screening to Personalised Risk
One of the most significant conceptual shifts underway is the move toward personalised screening strategies.
Traditional breast screening has largely been age-based and population-driven. The emerging model is risk-adapted screening, incorporating:
- Breast density
- Family history and genetics
- Prior imaging findings
- Lifestyle and hormonal factors
- AI-derived imaging biomarkers
This approach aims to stratify patients more effectively—intensifying surveillance for higher-risk individuals while potentially reducing unnecessary imaging in lower-risk groups.
While evidence is still evolving, national screening programmes are actively exploring how to operationalise this shift without increasing inequality or complexity.
Workflow Innovation: The Hidden Driver of Change
Much of the most impactful innovation in breast imaging is not visible in the images themselves, but in how they are processed.
Radiology workflows are being reshaped by:
- AI-assisted triage systems
- Structured reporting tools
- Automated measurement and annotation
- Integrated PACS/RIS platforms with embedded analytics
These developments are not just efficiency tools—they are responses to a structural imbalance between imaging demand and specialist workforce capacity.
The most successful implementations so far are those that treat AI and automation as workflow infrastructure, rather than standalone software.
Evidence, Adoption, and the Reality Gap
Despite rapid innovation, a consistent theme across the field is the gap between technological promise and real-world adoption.
Key tensions include:
- High-quality evidence versus early commercial deployment
- Pilot success versus scalable implementation
- Innovation speed versus regulatory caution
- Automation potential versus clinical accountability
As a result, breast imaging today sits in a hybrid state: mature in its imaging foundations, rapidly evolving in its analytical capabilities, but still consolidating its evidence base for widespread transformation.
The Road Ahead
Looking forward, several trajectories are becoming clearer:
- AI will become a standard layer within screening and diagnostic systems rather than a separate tool
- Risk-based screening models will gradually expand, particularly in organised national programmes
- Multimodal imaging interpretation will become more unified and algorithm-supported
- Workflow automation will be as important as diagnostic innovation
- Clinical governance frameworks will increasingly define the pace of adoption
Conclusion
Breast imaging innovation is no longer defined by isolated breakthroughs. It is defined by system-wide change—across technology, workflow, and clinical thinking.
The field is moving toward a model where imaging is not just about detecting disease earlier, but about continuously managing risk, integrating data, and supporting clinical decision-making at scale.
The state of innovation today is therefore best described not as a destination, but as a transition: from traditional imaging pathways toward a more intelligent, adaptive, and interconnected model of breast health care.